- info@buhlmannlabs.ch
- +41 61 487 12 12
- Home
- Products & Solutions
- Gastroenterology

CALPROTECTIN - The ideal tool to diagnose and monitor IBD patients
Calprotectin is the best marker for IBD. It is an established measure to discriminate IBD from IBS and a very valuable tool to monitor the disease course of IBD patients (Crohn’s and ulcerative colitis). BÜHLMANN Laboratories offers the largest product portfolio in the market of calprotectin testing and has set the standard for more than 10 years.

CALPROTECTIN - The ideal tool to diagnose and monitor IBD patients
Calprotectin is the best marker for IBD. It is an established measure to discriminate IBD from IBS and a very valuable tool to monitor the disease course of IBD patients (Crohn’s and ulcerative colitis). BÜHLMANN Laboratories offers the largest product portfolio in the market of calprotectin testing and has set the standard for more than 10 years.
Calprotectin
Calprotectin is the best marker for IBD. It is an established measure to discriminate IBD from IBS and a very valuable tool to monitor the disease course of IBD patients (Crohn’s and ulcerative colitis). The technology platforms offered by BÜHLMANN range from the gold standard ELISA assay, BUHLMANN fCAL®, to the POC test, Quantum Blue®, to the new IBDoc® home test and to the automated random access BÜHLMANN fCAL® turbo assay. With this portfolio all possible applications for measuring calprotectin are covered from the patient’s home test to the high throughput lab.
Therapeutic Drug Monitoring
Over the past two decades great improvements in therapy of chronic inflammatory diseases have been made. The rise of TNFα biologics like adalimumab and infliximab has been a great step forward to ameliorating disease course and keep inflammations at remission levels for prolonged periods of time. Patients with suboptimal drug concentrations have worse outcomes than those with adequate drug levels. Therapeutic drug monitoring (TDM) for adalimumab and infliximab has a great potential for the management of anti TNF therapy.
Quantum Blue®
The Quantum Blue® calprotectin rapid test is a sensitive, non-invasive tool to pre-select patients requiring colonoscopy for confirmation of organic bowel disease (IBD). Quantitative results for calprotectin concentration (in ug/g stool) are available within 12 minutes and are very comparable to the established BÜHLMANN fCAL® ELISA.
IBDoc®
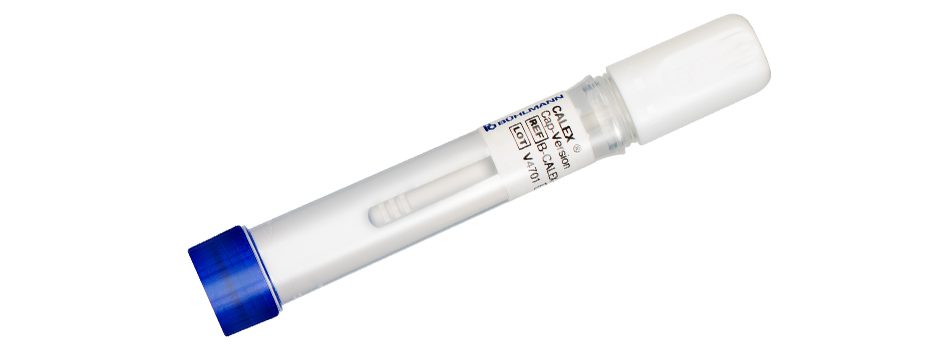
IBDoc® is the first in-vitro diagnostic home testing device measuring the inflammatory marker fecal calprotectin at home. The CalApp® turns your smartphone into a test cassette reader using state of the art image processing. Stool preparation is performed using the Calex® Valve that is characterized by its simple and convenient handling of stool samples. The secure connection of CalApp® with IBDoc® Portal allows Health Care Professionals to directly monitor patient results.
Automation
Immunoturbidimetry allows fast, flexible and random access applications with high precision. BÜHLMANN offers immunoturbidimetric solutions for the two gastroenterological markers fecal calprotectin and pancreatic elastase. The combination of both tests with the CALEX® Cap stool preparation device provides the best possible degree of automation for quantitative stool analysis.
CALEX® Cap
CALEX® Cap stool preparation device offers an efficient, convenient and hygienic preparation of stool samples. Its simple design makes it an ideal preparation device characterized not only by high safety but also by offering high stability of stool samples.

CALEX® Cap Collection Set
The CALEX® Cap stool preparation device offers a very useful solution when it comes to handling samples for fecal calprotectin and pancreatic elastase, outsourcing the collection step of stool to the patient. A simple to use patient kit that includes a buffer filled CALEX® Cap tube containing viricide and bactericide components, stool collection sheets, a user guide and plastic bag with patient label, allows for the sample to be safely sent or brought to lab collection sites and hospitals for further logistics and analysis.





Social Links